Peer-Reviewed Publications
-
Schmidt, E.; Vik, R.; Brubaker, B.; Abdulahad, D.; Monjure, T.; Battle, C.H.; Jayawickramarajah, J. Increasing Student Interest and Self-Efficacy in STEM by Offering a Service-Learning Chemistry Course in New Orleans. J. Chem. Ed. 2020 (Article ASAP).
-
Aryal, G.*; Battle, C. H.*; Grusenmeyer, T.; Zhu, M.; Jayawickramarajah, J. A Napthalimide Derived Fluorescence Sensor for Solid-Phase Screening of Cucurbit[7]uril-Guest Interactions. Chem. Comm. 2016 52, 2307-2310.
*These authors contributed equally to this work
-
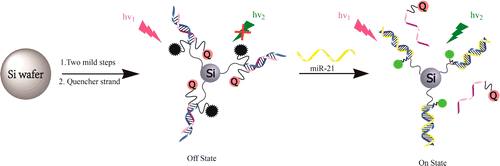
Chu, X.; Battle, C.; Zhang, N.; Aryal, G. H.; Madhusoodanan, M.; Jayawickramarajah, J. A Bile acid conjugated DNA chimera that conditionally inhibits carbonic anhydrase-II in the presence of microRNA-21. Bioconjugate Chem. 2015, 26 (8), 1606-1612.
-
Su, X.; Kuang, L.; Battle, C.; Shaner, T.; Mitchell, B. S.; Fink, M. J.; Jayawickramarajah, J., Mild Two-Step Method to Construct DNA-Conjugated Silicon Nanoparticles: Scaffolds for the Detection of MicroRNA-21. Bioconjugate Chem. 2014, 25 (10), 1739–1743.
-
Battle, C. H.; Chu, X.; Jayawickramarajah, J., Oligonucleotide-Based Systems for Input-Controlled and Non-Covalently Regulated Protein-Binding. Supramolecular Chemistry. 2013, 25 (12), 848-862.
-
Battle, C. H.; Jayawickramarajah, J., Supramolecular Approaches for Inhibition of Protein-Protein and Protein/DNA Interactions. In Supramolecular Chemistry: From Molecules to Nanomaterials, Steed, J. W.; Gale, P. A., Eds. John Wiley and Sons: 2012; Vol. 4, pp 1885-1908.
-
Mishur, R. J.; Griffin, M. E.; Battle, C. H.; Shan, B.; Jayawickramarajah, J., Molecular recognition and enhancement of aqueous solubility and bioactivity of CD437 by [beta]-cyclodextrin. Bioorg. Med. Chem. Lett. 2011, 21 (2), 857-860.
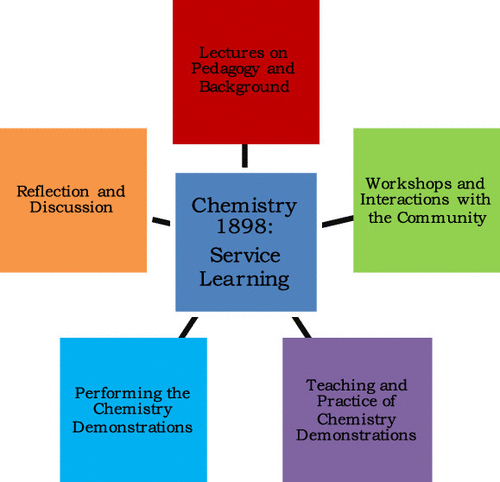
Increasing Student Interest and Self-Efficacy in STEM by Offering a Service-Learning Chemistry Course in New Orleans.
DOI: 10.1021/acs.jchemed.9b01140
There is a growing need for workers with STEM-aligned training in the modern global economy, but a paucity of workers to fill these positions. One important contributor to this issue is low student persistence in STEM. Nontraditional science courses that utilize more active-participation and learning are attractive as tools to increase student persistence and engender more interest in STEM. Herein is described the content and implementation of the undergraduate chemistry-based service-learning course, Chemistry 1898: Service Learning, that was offered in Spring 2019 at Tulane University. The goal of the course was to increase self-efficacy in chemistry and sustain undergraduate interest in STEM. The course also serves to increase STEM interest in the New Orleans public-school students. Chemistry 1898 features a well-rounded curriculum and diverse activities. The enrolled undergraduate students were not only taught chemistry concepts (general chemistry and supramolecular chemistry) but also asked to present the chemical concepts using attention-grabbing demonstrations to public-school students in the New Orleans area. In addition, the course covered multiple nonscience topics, including the pedagogy of service-learning, background on the New Orleans public-school system, and a guide for how to work with the community. The course also involved student reflection activities/surveys and interfaced with the Tulane Center for Public Service. Preliminary qualitative results from a set of anonymous pre- and poststudent surveys indicated that the undergraduate students gained self-efficacy in the general chemistry concepts covered in the course. Although the course did not have an effect on the career choices of the undergraduate students, the majority of the students were already very interested in a STEM career. Further, some students mentioned gaining a benefit in public speaking skills, and some considered the possibility of teaching and working with children in the future.
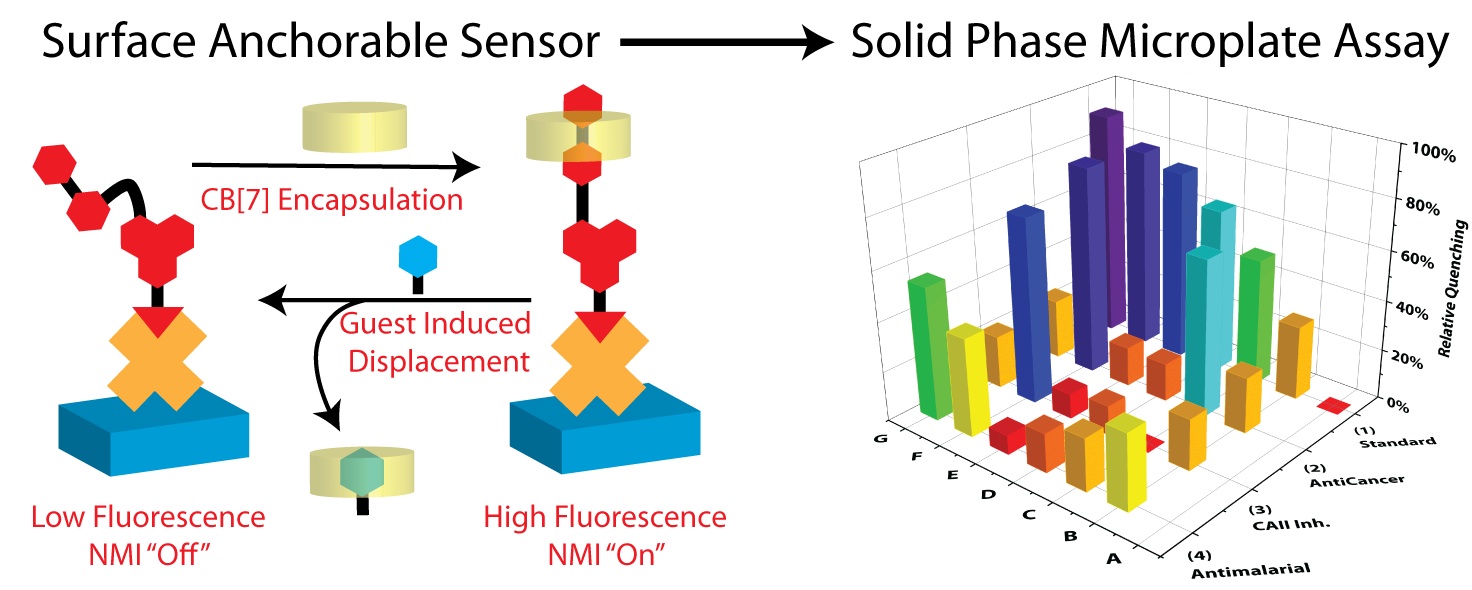
A naphthalimide derived fluorescent sensor for solid-phase screening of cucurbit[7]uril–guest interactions
A naphthalimide based fluorescent sensor displaying a significant increase in emission upon binding CB[7] with notable pH stability was developed and utilized in a surface-bound displacement assay for the rapid detection of CB[7] encapsulation of therapeutically relevant drug classes. Previously unknown binders with moderate to strong affinities were discovered.
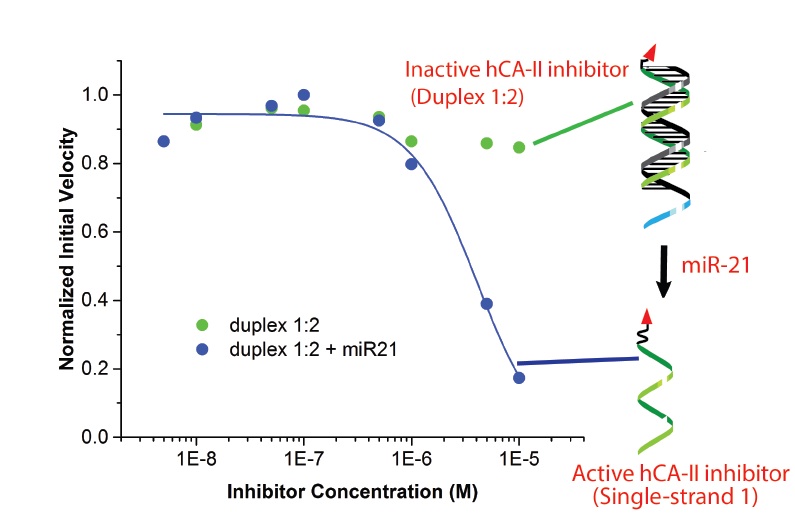
A Bile Acid Conjugated DNA Chimera that Conditionally Inhibits Carbonic Anhydrase-II in the Presence of MicroRNA-21
DOI: 10.1021/acs.bioconjchem.5b00231
In order to tackle the issue of systemic toxicity in chemotherapy, there is a need to develop novel mechanisms for the activation of protein inhibitors using biomarkers over-expressed in cancer cells. Many current strategies focus on using cancer associated enzymes as a triggering agent for prodrugs. Herein, we detail an alternative approach that harnesses a microRNA (miR-21) that is overexpressed in cancers as the trigger that activates an inhibitor of human carbonic anhydrase-II (hCA-II). Specifically, we have developed a DNA-small molecule chimera (DC) composed of an hCA-II binding lithocholic acid amide (LAA) head-group that can transition from a rigid duplex state (that does not bind appreciably to hCA) to a single stranded conformation via a miR-21 trigger. The activated single-stranded DC can project the LAA head-group into the hCA-II active site and is a robust hCA-II inhibitor (Ki of 3.12 μM). This work may spur research into developing new classes of cancer selective protein inhibitors.
Mild Two-Step Method to Construct DNA-Conjugated Silicon Nanoparticles: Scaffolds for the Detection of MicroRNA-21
We describe a novel two-step method, starting from bulk silicon wafers, to construct DNA conjugated silicon nanoparticles (SiNPs). This method first utilizes reactive high-energy ball milling (RHEBM) to obtain alkene grafted SiNPs. The alkene moieties are subsequently reacted with commercially available thiol-functionalized DNA via thiol–ene click chemistry to produce SiNP DNA conjugates wherein the DNA is attached through a covalent thioether bond. Further, to show the utility of this synthetic strategy, we illustrate how these SiNP ODN conjugates can detect cancer-associated miR-21 via a fluorescence ON strategy. Given that an array of biological molecules can be prepared with thiol termini and that SiNPs are biocompatible and biodegradable, we envision that this synthetic protocol will find utility in salient SiNP systems for potential therapeutic and diagnostic applications.
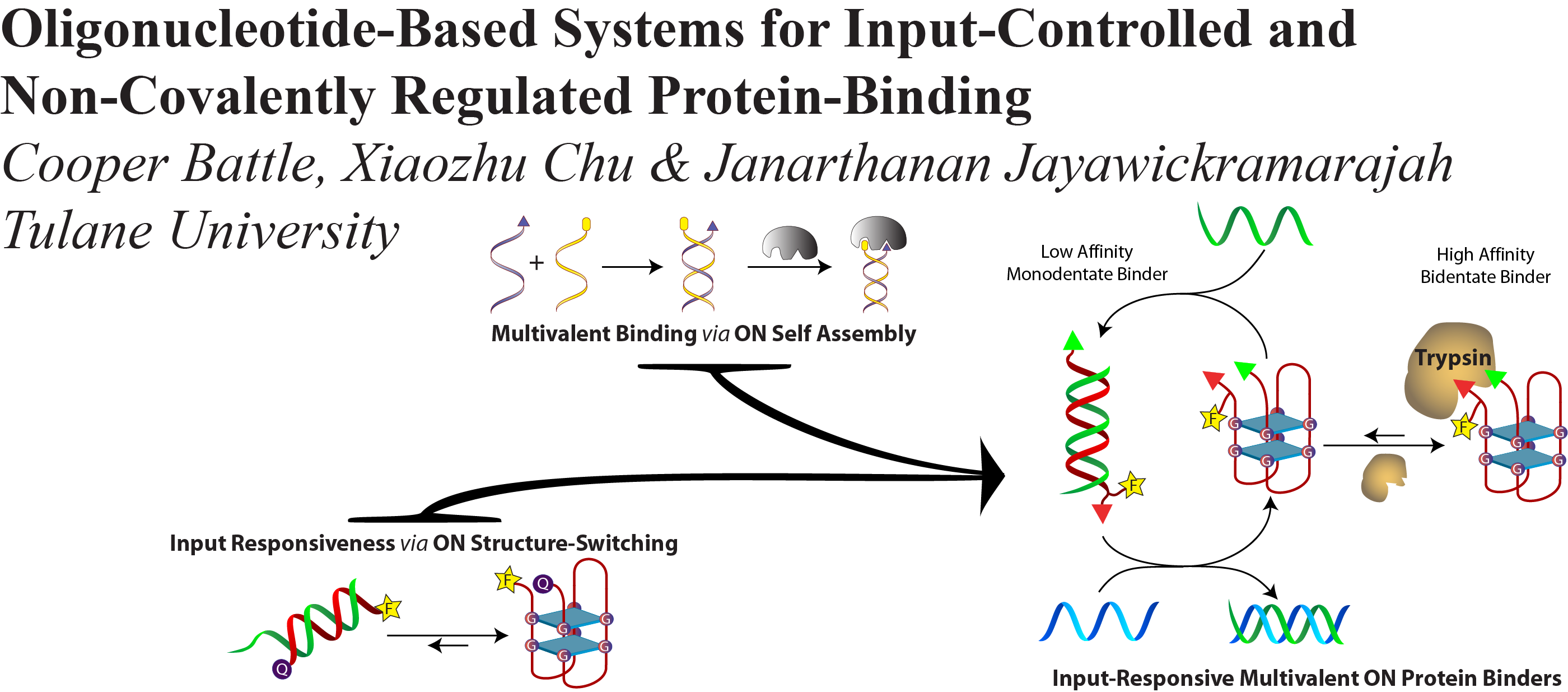
DOI: 10.1080/10610278.2013.810337
Supramolecular chemists continuously take inspiration from complex biological systems to develop functional molecules involved in molecular recognition and self-assembly. In this regard, ‘smart’ synthetic molecules that emulate allosteric proteins are both exciting and challenging, because many allosteric proteins can be considered as molecular switches that bind to other protein targets in a non-covalent fashion and, importantly, are capable of having their output activity controlled by prior binding to input molecules. This review discusses the foundations and passage towards the development of non-covalently operated oligonucleotide-based systems with protein-binding capacity that can be precisely regulated in an input-controlled manner.
Supramolecular Approaches for Inhibition of Protein–Protein and Protein–DNA Interactions
Found in: Supramolecular Chemistry: From Molecules to Nanomaterials, Volume 4
The disruption of protein–protein and protein–DNA interactions is increasingly being realized as important avenues for therapeutic intervention. Unlike the successes observed in the more mature field of small molecule derived active site antagonists, however, the sequestration of the sizeable, solvent exposed, and largely featureless protein surfaces remains a significant challenge. This chapter will provide a detailed account of the types of novel inhibitors being developed that address this challenge, with special emphasis given to supramolecular approaches. Indeed, while still in its infancy, the field of aqueous supramolecular chemistry has made great strides in the development of both covalent and self-assembled multivalent scaffolds for use in protein surface binding. While the former can provide rigid covalent scaffolds tailored for multivalent binding, the latter has the added benefit of being dynamic and thereby can rapidly lead to protein binders that allow for template-driven elucidation, are stimuli-responsive, and able to project heteromeric recognition elements. Taken together, these properties give supramolecular inhibitors the potential for exciting future applications in the disruption of protein interactions.
Molecular recognition and enhancement of aqueous solubility and bioactivity of CD437 by β-cyclodextrin
DOI: 10.1016/j.bmcl.2010.11.073
CD437 (6-[3-(1-adamantyl)-4-hydroxyphenyl]-2-naphthalene carboxylic acid) is a novel synthetic retinoic acid derivative that has been shown to selectively induce apoptosis in human lung cancer cells. This compound, however, is limited in its application due to its low solubility in aqueous solutions. One technique for increasing the solubility and bioavailability of a cytotoxic agent is the formation of inclusion complexes with cyclodextrins. Herein, we report the formation and characterization of a 2:1 complex between β-cyclodextrin (β-CD) and CD437. It is shown that CD437 is a tight binder of β-CD with an overall association constant of 2.6 ± 0.6 × 107 M−2. In addition, we demonstrate (a) that β-CD-derived complexation enhances the aqueous solubility of CD437, and (b) that a significant increase in the toxicity of CD437 against a human lung adenocarcinoma cell line can be achieved by co-treatment with β-CD.